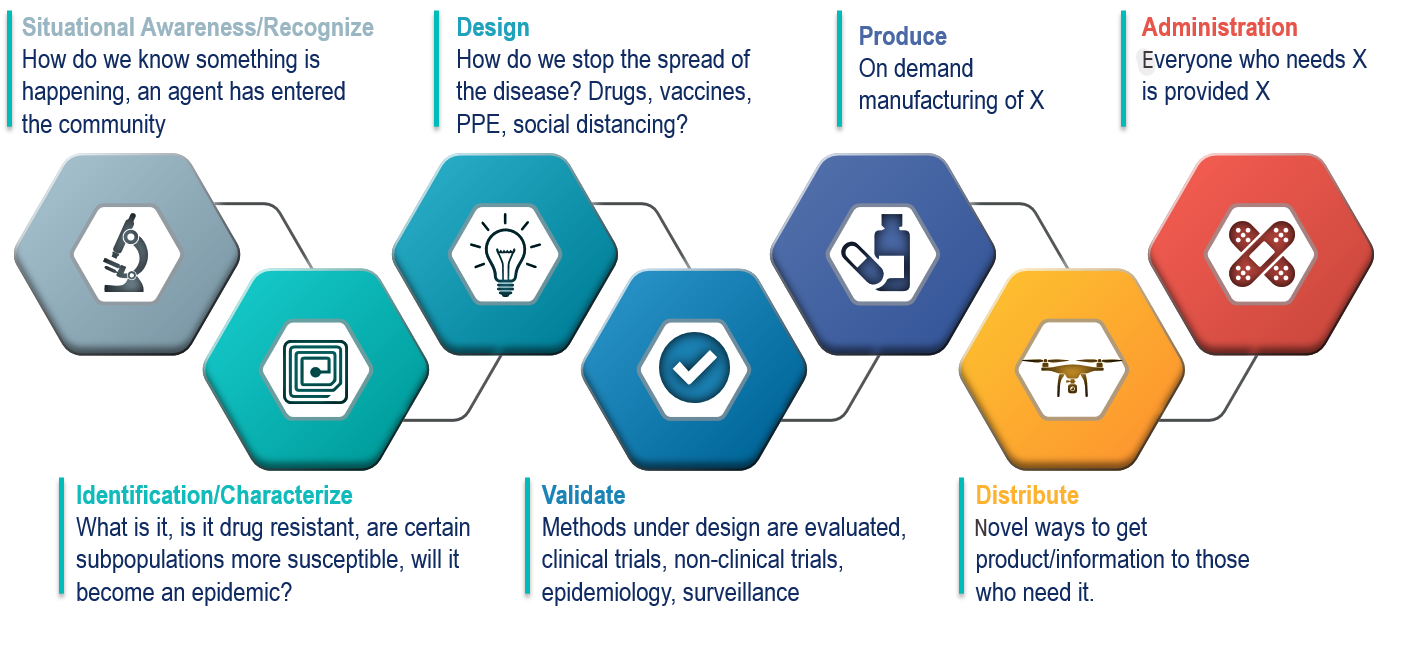
The Drive Business Model
DRIVe is forming unique public-private partnerships. DRIVe will leverage new authorities given to BARDA under the 21st Century Cures Act to stimulate innovation through both non-dilutive funding and dilutive funding. In addition, through our DRIVe Accelerator Network, we will have teams forward deployed throughout the USA to identify promising solutions wherever innovation is happening. Together we are building out an ecosystem of restless innovation, driven by industry and the entrepreneurial community, to address our nation's largest health security threats.